Our study of molecular reactivity has two dominant strands.
(1) Understanding how unusual chemistry occurs. In this area we have taken a leading role in explaining phenomena that is difficult to understand through classical mechanistic thinking. Once example of this approach is the work that we have been collaborating with the Murphy lab on to explore the use and development of super electron donors.
Understanding reactivity allows us to successfully tackle the second area of research in the molecular reactivity theme, which is the design of new reactive species. Within this area we have been active in the design of new super electron donors, new organic catalysts and organometallic catalysts with selected properties.
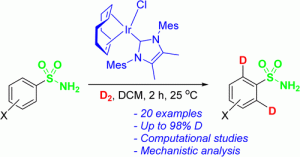
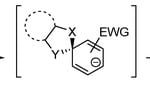
(2) Catalyst development has been an active area of research for the group over many years and we have experience working with Fe, Ru, Ir, and mixed-metal catalysts. An area in which we have had recent significant success is the tuning of the Ir deuteration catalysts from the Kerr group to expand substrate scope and regioselectivity of this class of catalysts. The selective deuteration of substrates is important for studying the activity and lifetime of these substrates in pharmaceutical applications.
Recent Reactivity Publications
-
 Adv. Synth. Catal. 2024 doi.org/10.1002/adsc.20240 [...]
Adv. Synth. Catal. 2024 doi.org/10.1002/adsc.20240 [...] -
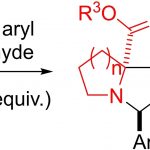 Chem. Eur. J. 2023, e202301861, 10.1002/chem.20230 [...]
Chem. Eur. J. 2023, e202301861, 10.1002/chem.20230 [...] -
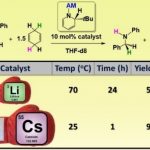 Angew. Chem. Int. Ed., 2023, e202304966, 10.1002/a [...]
Angew. Chem. Int. Ed., 2023, e202304966, 10.1002/a [...] -
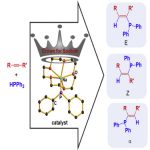 Cell Reports Physical Science., 2022, 100942
Cell Reports Physical Science., 2022, 100942