Theoretical Determination and Analysis of Chemical Properties
Understanding the relationship between the chemical structure and the observable experimental properties of a molecule, or system of molecules is critical to the design of systems that have tailored properties.
 Work within the TuttleLab has covered the calculation of a range of properties. This includes the calculation of Mössbauer parameters for iron-centered corroles was a study that led to the determination of the spin-state of these compounds through the calculation of their electronic structure and the corresponding correlation between the experimental and theoretical Mössbauer parameters
Work within the TuttleLab has covered the calculation of a range of properties. This includes the calculation of Mössbauer parameters for iron-centered corroles was a study that led to the determination of the spin-state of these compounds through the calculation of their electronic structure and the corresponding correlation between the experimental and theoretical Mössbauer parameters
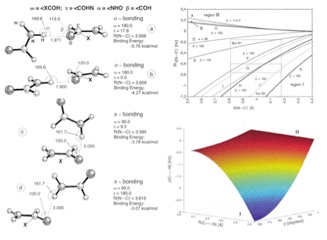
The development of a new method for analyzing spin-spin coupling constants (SSCCs) – the J-OC-PSP (Decomposition of J into Orbital Contributions using Orbital Currents and Partial Spin Polarisation) analysis method. Through the decoding of the SSCC this method is able to produce empirical relationships as well as an understanding of how the two properties (SSCC and geometry) are related. This method was used to study: (a) long-range coupling; (b) the through-space mechanism of SSCCs; and (c) the nature of the H-bond.
Recent work has focused on the utility of employing semi-empirical methods to monitor the dynamic evolution of NMR chemical shifts on the sub-picosecond timescale, which revealed the strong correlation between vibrational modes and the calculated chemical shifts in a heptapeptide.
The absorption spectra for organic dyes has also been studied with a focus on the applicability of the TD-DFT method for predicting the spectra of these compounds and the influence of solvent on the calculated absorption maxima.
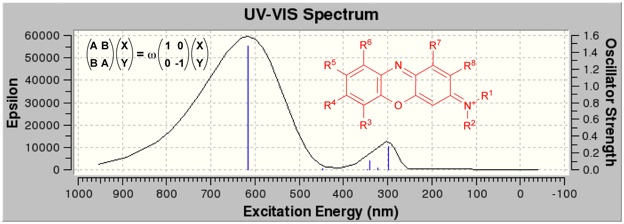
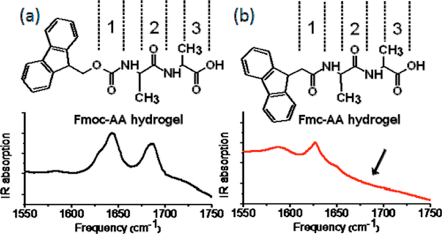
Finally the relationship between supramolecular structure and FTIR spectra and/or UV-Vis spectra for peptide-based self-assembled structures has become an increasingly active Rea of research.
Recent Property Publications
-
 J. Comput. Chem., 2023, 44, 1795.
J. Comput. Chem., 2023, 44, 1795. -
 Chemistry, Molecular Sciences and Chemical Enginee [...]
Chemistry, Molecular Sciences and Chemical Enginee [...] -
 Angew. Chem. Int. Ed., 2023, 62, e202218067
Angew. Chem. Int. Ed., 2023, 62, e202218067